If you're interested in becoming a contributor or requesting changes then click here to join the discord
Difference between revisions of "Nalu Medical"
Landonodnal (talk | contribs) |
Landonodnal (talk | contribs) |
||
| Line 25: | Line 25: | ||
! Device ID !! Device Name !! Class !! Category !! PMA !! PMN | ! Device ID !! Device Name !! Class !! Category !! PMA !! PMN | ||
|- | |- | ||
| − | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K202274 K202274] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True | + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K202274 K202274] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True |
|- | |- | ||
| − | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K190960 K190960] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True | + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K190960 K190960] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True |
|- | |- | ||
| − | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K221376 K221376] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True | + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K221376 K221376] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True |
|- | |- | ||
| − | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K203547 K203547] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True | + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K203547 K203547] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True |
|- | |- | ||
| − | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K201618 K201618] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True | + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K201618 K201618] || Stimulator, Spinal-Cord, Implanted (Pain Relief) || 2 || Neurology || False || True |
|} | |} | ||
| + | |||
| + | ==Hardware== | ||
| + | *[[BrainScope|BrainScope]] | ||
==Links== | ==Links== | ||
Latest revision as of 22:57, 17 February 2024
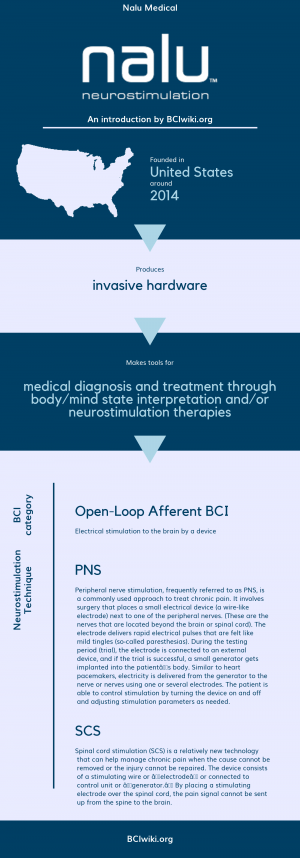
"Nalu Medical, Inc. is a privately held early-stage medical device start-up company. Our team of seasoned entrepreneurs, engineers and scientists are developing the next generation of medical devices. Our novel devices will address a number of poorly treated clinical conditions."
Founded in The United States around 2014, Nalu Medical produces invasive hardware.
Nalu Medical makes tools for medical diagnosis and treatment through body/mind state interpretation and/or neurostimulation therapies.
BCI Categories: Open-Loop Afferent
Neurostimulation Technique(s): SCS, PNS
FDA
Nalu Medical has 8 medical devices registered with the FDA. Here are some of them:
| Device ID | Device Name | Class | Category | PMA | PMN |
|---|---|---|---|---|---|
| K202274 | Stimulator, Spinal-Cord, Implanted (Pain Relief) | 2 | Neurology | False | True |
| K190960 | Stimulator, Spinal-Cord, Implanted (Pain Relief) | 2 | Neurology | False | True |
| K221376 | Stimulator, Spinal-Cord, Implanted (Pain Relief) | 2 | Neurology | False | True |
| K203547 | Stimulator, Spinal-Cord, Implanted (Pain Relief) | 2 | Neurology | False | True |
| K201618 | Stimulator, Spinal-Cord, Implanted (Pain Relief) | 2 | Neurology | False | True |
Hardware
Links
Website Crunchbase LinkedIn GitHub Twitter FacebookInstagramYouTube