If you're interested in becoming a contributor or requesting changes then click here to join the discord
Difference between revisions of "NeuraLace Medical"
Landonodnal (talk | contribs) |
Landonodnal (talk | contribs) |
||
| (8 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
| + | [[Category:GitHub Accounts]] | ||
| + | [[Category:LinkedIn Accounts]] | ||
| + | [[Category:Instagram Pages]] | ||
| + | [[Category:Facebook Pages]] | ||
| + | [[Category:Twitter Accounts]] | ||
[[Category:Organizations]] | [[Category:Organizations]] | ||
[[Category:Companies]] | [[Category:Companies]] | ||
| − | Founded in 2016, NeuraLace is a technology company with a focus on medical innovations to help to deal with nerve damage. The company is developing a non-invasive interface, bridging the gap between technology and neurology, to deliver sustainable chronic pain relief at scale (they do this with magnetic stimulation) . Currently, they have an Axon therapy product that uses this magnetic stimulation to target people with post-traumatic peripheral nerve pain (typically numbness in the foot and hand extremities). | + | "Founded in 2016, NeuraLace is a technology company with a focus on medical innovations to help to deal with nerve damage. The company is developing a non-invasive interface, bridging the gap between technology and neurology, to deliver sustainable chronic pain relief at scale (they do this with magnetic stimulation) . Currently, they have an Axon therapy product that uses this magnetic stimulation to target people with post-traumatic peripheral nerve pain (typically numbness in the foot and hand extremities)." |
| + | |||
| + | |||
| + | [[File:NeuraLace_Medical.png|thumb|NeuraLace Medical Company Profile]] | ||
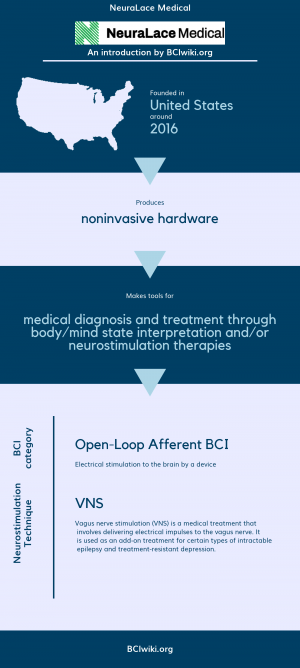
| + | Founded in The United States around 2016, NeuraLace Medical produces noninvasive hardware. | ||
| + | |||
| + | NeuraLace Medical makes tools for medical diagnosis and treatment through body/mind state interpretation and/or neurostimulation therapies. | ||
| + | |||
| + | [[Brain Computer Interface Classification|BCI Categories]]: Open-Loop Afferent | ||
| + | |||
| + | [[:Category:Neurostimulation_Techniques|Neurostimulation Technique(s)]]: VNS | ||
| + | ==FDA== | ||
| + | NeuraLace Medical has 1 medical devices registered with the FDA. Here are some of them: | ||
| + | {| class="wikitable" style="margin:auto" | ||
| + | |+ Examples of FDA Approved Devices ([[NeuraLace_Medical_FDA_Devices | View List]]) | ||
| + | |- | ||
| + | ! Device ID !! Device Name !! Class !! Category !! PMA !! PMN | ||
| + | |- | ||
| + | | [https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?db=pmn&id=K210021 K210021] || Electromagnetic Stimulator, Pain Relief || 2 || Neurology || False || True | ||
| + | |} | ||
| + | |||
| + | ==Hardware== | ||
| + | *[[BrainScope|BrainScope]] | ||
==Links== | ==Links== | ||
| + | |||
[http://www.neuralacemedical.com/ Website] | [http://www.neuralacemedical.com/ Website] | ||
[https://www.crunchbase.com/organization/neuralace-medical Crunchbase] | [https://www.crunchbase.com/organization/neuralace-medical Crunchbase] | ||
[https://www.linkedin.com/company/neuralace/ LinkedIn] | [https://www.linkedin.com/company/neuralace/ LinkedIn] | ||
[https://github.com/NeuraLaceMedical GitHub] | [https://github.com/NeuraLaceMedical GitHub] | ||
| + | [https://twitter.com/Neuralace?ref_src=twsrc%5Egoogle%7Ctwcamp%5Eserp%7Ctwgr%5Eauthor Twitter][https://facebook.com/neuralace/ Facebook][https://instagram.com/neuralacemedical/ Instagram] | ||
Latest revision as of 22:57, 17 February 2024
"Founded in 2016, NeuraLace is a technology company with a focus on medical innovations to help to deal with nerve damage. The company is developing a non-invasive interface, bridging the gap between technology and neurology, to deliver sustainable chronic pain relief at scale (they do this with magnetic stimulation) . Currently, they have an Axon therapy product that uses this magnetic stimulation to target people with post-traumatic peripheral nerve pain (typically numbness in the foot and hand extremities)."
Founded in The United States around 2016, NeuraLace Medical produces noninvasive hardware.
NeuraLace Medical makes tools for medical diagnosis and treatment through body/mind state interpretation and/or neurostimulation therapies.
BCI Categories: Open-Loop Afferent
Neurostimulation Technique(s): VNS
FDA
NeuraLace Medical has 1 medical devices registered with the FDA. Here are some of them:
| Device ID | Device Name | Class | Category | PMA | PMN |
|---|---|---|---|---|---|
| K210021 | Electromagnetic Stimulator, Pain Relief | 2 | Neurology | False | True |