If you're interested in becoming a contributor or requesting changes then click here to join the discord
Difference between revisions of "InsighTec"
Landonodnal (talk | contribs) |
Landonodnal (talk | contribs) |
||
| Line 2: | Line 2: | ||
[[Category:Companies]] | [[Category:Companies]] | ||
ExAblater 2000 system was the first system to use the MR guided focused ultrasound technology. The ExAblate system received CE mark in 2002 and the Food and Drug Administration (FDA) approval in 2004 for the treatment ofsymptomatic uterine fibroids. ExAblate received the European CE Mark for pain palliation of bone metastases in June 2007 and for adenomyosis in June 2010. This device has won numerous innovation awards and has been used to treat thousands of patients around the world. | ExAblater 2000 system was the first system to use the MR guided focused ultrasound technology. The ExAblate system received CE mark in 2002 and the Food and Drug Administration (FDA) approval in 2004 for the treatment ofsymptomatic uterine fibroids. ExAblate received the European CE Mark for pain palliation of bone metastases in June 2007 and for adenomyosis in June 2010. This device has won numerous innovation awards and has been used to treat thousands of patients around the world. | ||
| + | |||
| + | |||
| + | [[File:InsighTec.png|thumb|InsighTec Company Profile]] | ||
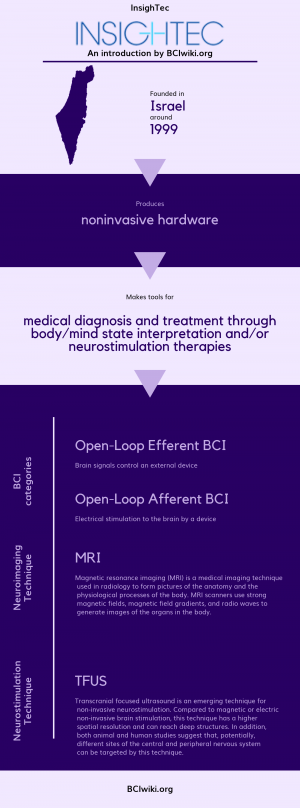
| + | Founded in Israel around 1999, InsighTec produces noninvasive hardware. | ||
| + | |||
| + | InsighTec makes tools for medical diagnosis and treatment through body/mind state interpretation and neurostimulation therapies. | ||
| + | |||
| + | [[Brain Computer Interface Classification|BCI Categories]]: Open-Loop Efferent, Open-Loop Afferent | ||
==Links== | ==Links== | ||
[http://www.insightec.com/ Website] | [http://www.insightec.com/ Website] | ||
[https://www.crunchbase.com/organization/insightec Crunchbase] | [https://www.crunchbase.com/organization/insightec Crunchbase] | ||
[https://www.crunchbase.com/organization/insightec LinkedIn] | [https://www.crunchbase.com/organization/insightec LinkedIn] | ||
Revision as of 02:09, 22 February 2022
ExAblater 2000 system was the first system to use the MR guided focused ultrasound technology. The ExAblate system received CE mark in 2002 and the Food and Drug Administration (FDA) approval in 2004 for the treatment ofsymptomatic uterine fibroids. ExAblate received the European CE Mark for pain palliation of bone metastases in June 2007 and for adenomyosis in June 2010. This device has won numerous innovation awards and has been used to treat thousands of patients around the world.
Founded in Israel around 1999, InsighTec produces noninvasive hardware.
InsighTec makes tools for medical diagnosis and treatment through body/mind state interpretation and neurostimulation therapies.
BCI Categories: Open-Loop Efferent, Open-Loop Afferent