If you're interested in becoming a contributor or requesting changes then click here to join the discord
Difference between revisions of "Synergia Medical"
Landonodnal (talk | contribs) |
Landonodnal (talk | contribs) |
||
| Line 1: | Line 1: | ||
| + | [[Category:LinkedIn Accounts]] | ||
[[Category:Organizations]] | [[Category:Organizations]] | ||
[[Category:Companies]] | [[Category:Companies]] | ||
Latest revision as of 01:50, 12 April 2022
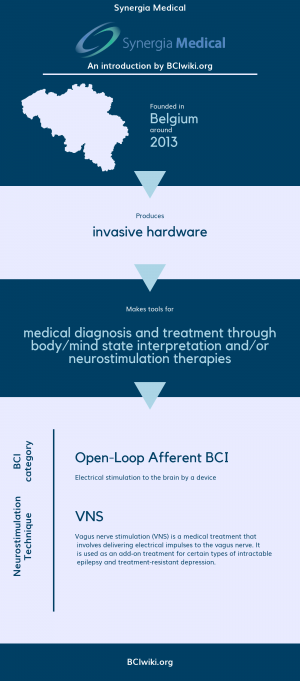
Synergia Medical aims at developing and commercializing next generation neurostimulation devices. It relies on a seasoned management team with extensive experience in developing implantable neurostimulators. In only three years time, the company became ISO13485 certified, created and sold an OEM external neurostimulator for a motorized amputee prosthesis arm and developed its first version of the NAOS platform. Supported by private investors and public research grants, the company has quickly grown to a team of thirteen highly skilled multidisciplinary professionals.
Founded in Belgium around 2013, Synergia Medical produces invasive hardware.
Synergia Medical makes tools for medical diagnosis and treatment through body/mind state interpretation and/or neurostimulation therapies.
BCI Categories: Open-Loop Afferent
Neurostimulation Technique(s): VNS